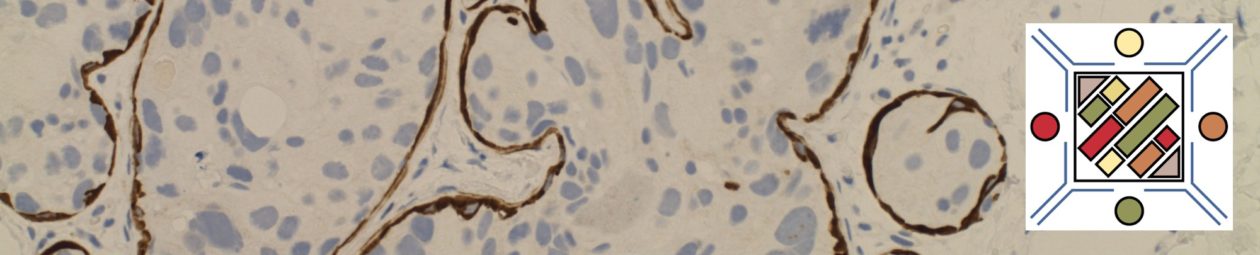
One of the most important aspects of breast cancer diagnosis is the evaluation of therapeutic markers (ER, PR, and HER2). Ki-67 is often included in the panel as a prognostic marker. ER expression determines eligibility to receive hormonal therapy (Tamoxifin), PR expression is a prognostic marker, and HER-2 over-expression determines eligibility to receive Herceptin®. Diagnostically, the challenge is to consistently and accurately perform and interpret these IHC markers.
Estrogen Receptor (ER):
- Nuclear Marker
- Stain is reported as PERCENT STAINING OF TUMOR CELLS and STAIN INTENSITY (1+, 2+, 3+).
- 1% or greater nuclear expression in tumor cells is considered positive, and therefore eligible to receive hormonal therapy.
- CAP-ASCO recommendations are for <1 hr. from time of excision/biopsy to having a cut edge of tumor in 10% neutral bufferedormalin fixative. Fixation window of 6-72 hrs. These times should be noted in the pathology report (time of excision, time in gross room, and time in fixative).
- Negative staining results in biopsy material without an internal control should be repeated on the excisional specimen using blocks with both tumor and benign breast parenchyma.
Progesterone Receptor (PR):
- Nuclear Marker
- Stain is reported as PERCENT STAINING OF TUMOR CELLS and STAIN INTENSITY (1+, 2+, 3+).
- 1% or greater nuclear expression in tumor cells is considered positive.
- PR expression is a prognostic marker, and not directly used for eligibility to receive a specific treatment.
- PR expression without ER expression should raise significant concern that the ER and PR slides have been mixed up, or there is a problem with the ER assay. Many scientists believe that ER expression is required for PR expression.
HER-2 Overexpression (HER-2):
- Membraneous stain
- Stain is interpreted by combining stain intensity and percentage of tumor involvement to classify as (0, 1+, 2+, or 3+).
-
- 0 (negative) = No staining or cell membrane staining in <10% of tumor cells.
- 1+ (negative) = Faint membrane staining (partial membrane staining) in >10% of tumor cells.
- 2+ (equivocal) = Weak to moderate complete membrane staining in >10% of tumor cells, or strong complete staining in <10% of invasive tumor cells.
- 3+ (positive) = Strong complete membrane staining in >10% of tumor cells.
- CAP-ASCO recommendations are for <1 hr. from time of excision/biopsy to having a cut edge of tumor in 10% neutral buffered fomalin fixative. Fixation window of 6-72 hrs. Over-fixation is probably not a clinically significant issue practically, but given the absence of relevant IHC data and the highly regulated environment surrounding HER2 testing, f/u FISH testing for negative results (outside the fixative window) is necessary.
- Equivocal (2+) results should be followed-up with FISH testing, if IHC is used as the initial testing modality (most common). Less than 1/3rd of equivocal cases show Her2 over-expression by FISH analysis.
References
Hammond ME, et. al. “ASCO-CAP Guideline Recommendations for IHC Testing of ER and PR in Breast Cancer”. Arch Pathol Lab Med-Vol. 134, June 2010.
Wolff, A. C., Hammond, M. E. H., Hicks, D. G., Dowsett, M., McShane, L. M., Allison, K. H., et al. (2013). Recommendations for Human Epidermal Growth Factor Receptor 2 Testing in Breast Cancer: American Society of Clinical Oncology/College of American Pathologists Clinical Practice Guideline Update. Archives of pathology & laboratory medicine. doi:10.5858/arpa.2013-0953-SA
Tafe, L. J., Janjigian, Y. Y., Zaidinski, M., Hedvat, C. V., Hameed, M. R., Tang, L. H., et al. (2011). Human epidermal growth factor receptor 2 testing in gastroesophageal cancer: correlation between immunohistochemistry and fluorescence in situ hybridization. Archives of pathology & laboratory medicine, 135(11), 1460–1465. doi:10.5858/arpa.2010-0541-OA
Arch Pathol Lab Med. 2001;125:746.